Research Article
Hiatal Hernioplasty with a Novel Composite Mesh Design: Results of a Multicenter Study after Two- Years Follow-up
Delgado Fernando1, Aguiló Javier2, Asencio Francisco3, Primo Vicent4, Gomez-Abril Segundo1, Saborit Rosa2 and Carvajal-López Fernando3*
1Department of Surgery, Hospital Universitario Doctor Peset, Spain
2Department of Surgery, Hospital Público Lluis Alcanyis De Xátiva, Spain
3Department of Surgery, Hospital Arnau de Vilanova, Spain
4Department of Surgery, Hospital de Dénia-MarinaSalud, Spain
*Corresponding author: Fernando Carvajal López, Department of Surgery, Hospital Arnau de Vilanova, Carrer Sant Climent, 12, 46015, Valencia, Spain
Published: 17 Oct, 2018
Cite this article as: Fernando D, Javier A, Francisco A,
Vicent P, Segundo G-A, Rosa S, et
al. Hiatal Hernioplasty with a Novel
Composite Mesh Design: Results of
a Multicenter Study after Two- Years
Follow-up. World J Surg Surgical Res.
2018; 1: 1064
Abstract
Background: Surgical treatment of paraesophageal hiatal hernia is still controversial, particularly
in aspects related to surgical technique, being the main problem the high recurrence rate, between
20% and 30%.
Methods: The aim of this study is to describe the surgical technique and results of laparoscopic
repair of paraesophageal hernia using a new composite silicone/polypropylene mesh. This is a
retrospective study of 61 patients who underwent laparoscopic repair of symptomatic large (6 cm
or more in diameter) hiatal hernias in three public hospitals of the Valencia Community (Spain).
The study analyzes postoperative mesh-related morbidity, functional outcomes and recurrence rate.
Results: There were two conversions due to technical difficulties. The mean hospital stay was 3.2
days. Mean follow-up was 26 months. A recurrence rate of 11.7% was recorded. Complications,
reoperations and quality of life are analyzed.
Conclusion: This newly designed prosthetic mesh offers easy laparoscopic implantation with
acceptable recurrence and complications. Additional studies are needed to determine the most
appropriate material and shape to cover hiatal defects.
Keywords: Paraesophageal hernia; Hiatoplasty; Hiatal hernioplasty; Mesh; Laparoscopic repair
Introduction
Hiatal Hernia (HH) is a common disorder characterized by enlargement of the space between
the diaphragmatic crura, secondary to increased intra-abdominal. Current classification of HH
defines 4 types of hiatal or Paraesophageal Hernias (PEH): Type 1: Sliding hernia; Type 2: True PEH
or rolling hernia, herniation of gastric funds with gastroesophageal junction in the normal anatomic
location; Type 3: Combination of types 1 and 2 a Type 4: Herniation of other intraabdominal viscera
[1].
Large hiatal defects (>6 cm) may cause giant hernias that are associated with symptoms like
chest pain, vomit and post-prandial dysphagia and may lead to torsion, perforation and massive
bleeding [2].
Surgical treatment of paraesophageal hiatal hernia is still controversial, particularly in aspects
related to surgical technique and functional outcomes [3-5]. Laparoscopic repair of paraesophageal
hernia is more complex than that of GERD, although it offers similar benefits when performed by
experienced surgeons, with less pain, better recovery and shorter hospital stay with no increase in
morbidity [6].
The main problem with surgical treatment is still the high recurrence rate, between 20% and 30%.
Although not always associated with symptoms, recurrence requires redo operations in a significant
number of patients and has been attributed to excessive tension after the closure of large hiatal
defects [7]. To overcome this problem, meshes were introduced to provide tensionless cover of such
defects [8-10]. The meshes initially used were made of non-absorbable materials (polypropylene and
PTFE) as used for inguinal hernia repair. Serious complications have been reported, particularly
with polypropylene meshes, related to fibrosis thus conditioning dysphagia and even esophageal erosion [11-13]. Subsequently, absorbable biological meshes have been used to avoid this situation. Mesh repairs show a decrease in
recurrence when compared with primary closure in several studies
[14-18].
The aim of this study is to describe the surgical technique and
results of laparoscopic repair of paraesophageal hernias using a
new composite silicone/polypropylene mesh. The study analyzes
postoperative mesh-related morbidity, functional outcomes and
recurrence rate.
Methods and Materials
Patients
This is a retrospective study of 61 patients who underwent
laparoscopic repair of symptomatic paraesophageal hiatal hernias
(types II, III and IV) in three public hospitals of the Valencian
Community (Spain) (Dr. Peset Hospital in Valencia, Lluís Alcanyís
Hospital in Xativa and Arnau de Vilanova Hospital in Valencia).
The study analyzes operative morbidity, anatomic recurrence,
reoperation, mesh-related complications and quality of life by means
of the Visick scale. In a subset of patients belonging to one of the
participating centers the GIQLI test (Gastrointestinal Quality of Life
Index) was also used [14].
Diagnostic work-up
All patients underwent clinical assessment, barium swallow and
gastroscopy. CT scan was indicated in cases of complex hernias (more
than one organ) or gastric volvulus. When gastroesophageal reflux
disease or an esophageal motor disorder was suspected, esophageal
manometry and 24 hr pH-metry were carried out.
Surgical technique
All operations were performed by experienced surgeons in
laparoscopic and esophagogastric surgery.
A standardized 5-trocar laparoscopic approach was performed.
The procedure begins with a thorough dissection of the esophageal
hiatus, avoiding the vagus nerves and fully exposing the crura.
Herniated organs are reduced in the abdomen, the hernia sac is
resected and the esophagus is dissected free from its mediastinal
attachments to obtain at least 3 cm of intra-abdominal esophagus. The
hiatal defect is closed by approximating the diaphragmatic crura with
non-absorbable sutures in the retroesophageal space. A maximum of
two stitches are used to avoid kinking of the esophagus. Additional
sutures can be utilized to close the pre-esophageal gap of the defect if
necessary with a maximum of two.
Indications for mesh implantation: The use of a reinforcing
mesh is indicated when hiatal defects 6 cm or more in largest
diameter or excessive tension in the standard diaphragmatic suture
is encountered. A composite mesh designed by the first author
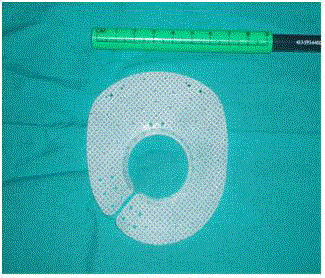
(MicroVal-France) is subsequently placed (Figure 1).
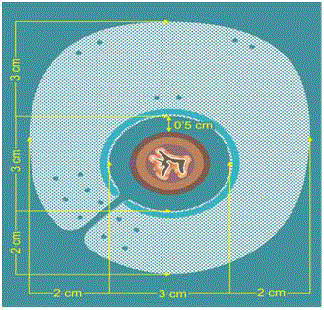
The mesh consists of two layers, polypropylene and silicone, for
the diaphragmatic and peritoneal sides respectively, measuring 7 cm
× 8 cm in size with an adjustable 3 cm central ring, opened at the
right lower rim to allow passage of the esophagus. Furthermore, the
ring has a silicone reinforcement to prevent erosion of the esophagus.
It can be easily inserted around the esophagus due to its preformed
shape. Once in place, the ring is closed with a suture including the
right crus leaving a 0.5 cm gap with the esophagus to avoid excessive
contact or migration of the fundus (Figure 2). An additional suture
needs to be attached at the opposite side of the hiatal orifice for proper
mesh fixation. Supplementary stitches or cyanoacrilate glue are used
when considered necessary. After the anatomic reconstruction a
short floppy 360° fundoplication is performed.
Follow-up
A clinical assessment was carried out to all patients at least
three months and one year after the operation. Special attention was
focused on dysphagia, pain or reflux symptoms. Recurrence was
systematically monitored by performing a barium meal at one year.
Figure 1
Results
Sixty one patients, 46 women and 15 men were included in
the study with a mean age of 66 years (range 46-84). Mean body
mass index was 29.70 (range 22.9-40). The ASA risk factor was
distributed as follows: 12% classified as ASA I, 65% as ASA II and
23% as ASA III. Predominant symptoms were heartburn in 53.5%
of the cases, dysphagia in 42.2%, epigastric or retrosternal pain in
41.7%, vomiting in 38.4%, regurgitation in 34.5%, and anemia in
20% and respiratory symptoms in 19.5%. All patients underwent
barium swallow and endoscopy, 39% had also a CT scan and 18%
esophageal pH measurement and manometry. All hernias contained
the stomach, two cases included greater omentum and transverse
colon was contained in one. In 4 cases an incidental opening of the
pleura occurred during the operation with no consequences for the patient. The average operative time was 116 mins (range 75-240).
There were two elective conversions due to intra-operative technical
difficulties (3.3%). Two patients (3.3%) had to be re-operated in
the first 24 hrs due to intra-abdominal hemorrhage in one case and
immediate recurrence with incarceration of the stomach in the other.
Oral intake was initiated during the first 24 hrs in 61% of patients. The
mean hospital stay was 3.2 days (range 2-14). There was no mortality
in our series.
The mean follow-up was 26 months (range 12-48). Three patients
were lost to follow-up (5%). Anatomical recurrence was evidenced
in 11.7% of the patients, although in half of these, the patients either
did not present symptoms or referred clinical improvement after the
operation. The Visick scale was tested in all cases, with a 52% of Visck
I, 39.5% of Visick II and 14.5% of Visick III patients. Two patients
needed esophageal dilatations due to postoperative dysphagia with
good final outcome. Five patients (8%) had to be reoperated, because
of severe symptomatic recurrence in three and intraluminal migration
of the mesh in two. In the subset of 15 patients where quality of life
was assessed by means of a GIQLI test, 10 patients had a score higher
than 106 (higher than the normal population), and 5 ranked below.
Discussion
Currently there is little controversy in gastro-esophageal reflux
surgery. The advantages of laparoscopic approach in terms of shorter
hospital stay, postoperative morbidity and improved quality of life
compared to open surgery has been documented, with no differences
in postoperative results [20,21]. 360° fundoplication seems to be the
option offering the lowest recurrence rate in the long-term when
compared to other techniques [22-24].
Paraesophageal hernias, which include hiatal hernias type II,
III and IV, are a different problem [25]. Its surgical treatment is
still controversial mainly in relation to elderly patients and patients
with comorbidity [26]. There is consensus in restricting surgery to
symptomatic patients with dysphagia, retrosternal pain, dyspnea,
dyspepsia, heartburn or anemia. Occasionally, emergency surgery
is needed to deal with complications such as incarcerated volvulus,
perforation, ischemia or bleeding. In the present study, all patients
were symptomatic. Regarding diagnosis, essential preoperative
studies are barium meal and upper endoscopy. CT can be useful in
emergency situations to detect complications caused by volvulus. If a
motor disorder is suspected, a ph-metry and esophageal manometry
should be performed [27].
Most authors agree in the advantage of laparoscopic approach,
the need for excision of the hernia sac and the restoration of the
gastroesophageal junction into the abdominal cavity [28]. The
association of an antire flux procedure is currently recommended over
simple anatomic reduction, due to the need of extensive dissection
of the hiatus, thus facilitating postoperative gastroesophageal reflux.
Nevertheless some studies do not support this point [29-31].
The main issue in surgical treatment of paraesophageal hernias
is the high recurrence rate as observed in all published series.
Recurrence does not always imply a bad clinical outcome since
many of these patients are no or mildly symptomatic [32,33]. This
was also our experience. An efficient closure of the hiatal defect is
the most determinant factor [34]. The use of reinforcing meshes
was initiated in the late 90’s with the use of polypropylene and
polytetrafluoroethylene materials as in abdominal wall repairs.
Severe adverse effects were reported with these materials, such as
mesh migration, esophageal erosion, fibrosis and stenosis, leading
to occasional esophageal and gastric resections. The placement of
reinforcing meshes in the hiatus generates controversies such as
true recurrence reduction, appropriate indication of the procedure
-defect's size vs. suture tension-; appropriate mesh attachment
-sutures vs. tacks vs. adhesives-; and proper esophageal protection
[35]. A reduction of the recurrence rate under 15% has been reported
with the use of meshes during the first years of follow-up, but a long
term decrease in recurrence has not been demonstrated yet [36-38].
In the present study, the recurrence rate was 11% after a mean
follow-up of 26 months. Some authors indicate the need of mesh
reinforcement when hiatal defects larger than 5 cm or 6 cm or
when excessive suture tension occurs [39]. It is known that nausea
and vomiting in the early postoperative period favor immediate
recurrence, as it occurred in one of our patients who had to be
reoperated in the first 24 hrs. There were two cases of mesh erosion and
subsequent migration into the gastric lumen that needed reoperation.
This complication could be attributed to excessive closure of the
mesh around the gastroesophageal junction. It is very important to
leave enough free space (0.5 cm) around the esophagus to permit
free mobility of the organ. The use of tissue adhesive combined with
sutures has been useful, facilitating the initial fixation of the mesh.
The use of tacks for fixation is discouraged due to the risk of injury
to nearby structures such as the aorta, the pericardium or the lung.
The most innovatory aspect of this study concerns the new type
of mesh used, made of composite, flexible, synthetic materials with an
original shape designed to avoid contact of the polypropylene layer
either with the esophagus or the peritoneal cavity. The peritoneal
side of the mesh and the esophageal circumferential rim are made
of silicone.
The characteristics of an ideal prosthesis for this anatomical
region should include easy handeling during laparoscopy, strong and
rapid tissue integration on the diaphragmatic surface, together with
minimal adherence to hollow viscera, minimal shrinkage, and good
transparency for secure fixation.
To cope with these principles our rationale was to use a double
layer mesh. A polypropylene side in order to reduce recurrence risk,
believing that polypropylene rapidly incorporates to tissue developing
a scar that strengthens the muscular fibers of the hiatal crura [40]
and a silicone side due to its low adhesive potential, believing that its
use at the hiatus might reduce the risk of erosive complications. We
preferred silicone to PTFE because of its known ‘‘mesh shrinkage’’
effect [41].
Silicone, the main compound used in this innovative prosthesis,
is an inert substance commonly used in breast implants since 1960.
The safety of the compound was approved in 2000 by FDA [42].
Since then many investigations have proved its safety in medical uses.
Considering the Cumberland criteria, silicone seems to be an ideal
substance to be used for repairing hernia [43].
In our literature review we have only found one study using a
silicone reinforced mesh for hiatal hernia repair in humans, with
good results [44].
We believe that the decrease in recurrence obtained in the present
study is due to the particular shape of the mesh, with its widest side
being anterior, thus reinforcing the widest and less muscular area of
the hiatal defect. The mesh presented has several openings in each branch facilitating its fixation with stitches to the crura and the
diaphragm. The internal circular silicone reinforcement of this mesh
allows free frictionless sliding of the esophagus, thus minimizing
the risk of erosion or intraluminal prosthesis migration. To our
knowledge this mesh design has not been previously described.
Our study presents the inherent constraints of a retrospective
series with a still limited follow-up, particularly in relation to
recurrence and quality of life, although 30% of the patients were
followed for more than 4 years with a satisfactory outcome. Additional
studies are needed to determine the most appropriate material and
shape to cover hiatus defects.
Figure 2
Disclosures
Dr. Delgado F, as designer of the prosthesis, has a commercial
relation with the manufacturer (Microval - France)
Drs. Aguiló J. Asencio F., Primo V. Saborit R., and Gomez-Abril
S. have no conflicts of interest or financial ties to disclose.
The manufacturing company has not sponsored or influenced by
any means in the free opinion of the authors.
References
- Oleynikov D, Jolley JM. Paraesophageal hernia. Surg Clin North Am. 2015;95(3):555-65.
- Romano A, D´Amore D, Esposito G, Petrillo M, Pezzella M, Romano FM, et al. Characteristics and outcomes of laparoscopic surgery in patients with large hiatal hernia. A single center study. Int J Surg Case Rep. 2018;48:142-4.
- Zhang C, Liu D, Li F, Watson DI, Gao X, Koetje JH, et al. Systematic review and meta-analysis of laparoscopic mesh versus suture repair of hiatus hernia: objective and subjective outcomes. Surg Endosc. 2017;31(12):4913-22.
- Memon MA, Memon B, Yunus RM, Khan S. Suture Cruroplasty versus Prosthetic Hiatal Herniorrhaphy for Large Hiatal Hernia: A Meta-analysis and Systematic Review of Randomized Controlled Trials. Ann Surg. 2016;263(2):258-66.
- Tam V, Winger DG, Nason KS. A systematic review and meta-analysis of mesh vs suture cruroplasty in laparoscopic large hiatal hernia repair. Am J Surg. 2016;211(1):226-38.
- Perdikis G, Hinder RA, Filipi CJ, Walenz T, McBride PJ, Smith S, et al. Laparoscopic paraesophageal hernia repair. Arch Surg. 1997;132(6):586-9.
- Mehta S, Boddy A, Rhodes M. Review of outcome after laparoscopic paraesophageal hiatal hernia repair. Surg Laparosc Endosc Percutan Tech. 2006;16(5):301-6.
- Granderath FA, Carlson MA, Champion JK, Szold A, Basso N, Pointner R, et al. Prosthetic closure of the esophageal hiatus in large hiatal hernia repair and laparoscopic antireflux surgery. Surg Endosc. 2006;20(3):367-79.
- Johnson JM, Carbonell AM, Carmody BJ, Jamal MK, Maher JW, Kellum JM, et al. Laparoscopic mesh hiatoplasty for paraesophageal hernias and fundoplications: a critical analysis of the available literature. Surg Endosc. 2006;20(3):362-6.
- Targarona E, Bendahan G, Balague C, Garriga J, Trias M. Mesh in the hiatus. A controversial issue. Arch Surg. 2004;139(12):1286-96.
- Stadlhuber RJ, Sherif AE, Mittal SK, Fitzgibbons RJ Jr, Michael Brunt L, Hunter JG, et al. Mesh complications after prosthetic reinforcement of hiatal closure: a 28-case series. Surg Endosc. 2009;23(6):1219-26.
- De Moor V, Zalcman M, Delhaye M, El Nakadi I. Complications of mesh repair in hiatal surgery: about 3 cases and review of the literature. Surg Laparosc Endosc Percutan Tech. 2012;22(4):e222-5.
- Oelschlager BK, Pellegrini CA, Hunter J, Soper N, Brunt M, Sheppard B, et al. Biologic Prosthesis Reduces Recurrence After Laparoscopic Paraesophageal Hernia Repair. A Multicenter, Prospective, Randomized Trial. Ann Surg. 2006;244(4):481-90.
- Schmidt E, Shaligram A, Reynoso JF, Kothari V, Oleynikov D. Hiatal hernia repair with biologic mesh reinforcement reduces recurrence rate in small hiatal hernias. Dis Esophagus. 2014;27(1):13-7.
- Jacobs M, Gomez E, Plasencia G, Lopez-Penalver C, Lujan H, Velarde D, et al. Use of surgisis mesh in laparoscopic repair of hiatal hernias. Surg Laparosc Endosc Percutan Tech. 2007;17(5):365-8.
- Andujar JJ, Papasavas PK, Birdas T, Robke J, Raftopoulos Y, Gagné DJ, et al. Laparoscopic repair of large paraesophageal hernia is associated with a low incidence of recurrence and reoperation. Surg Endosc. 2004;18(3):444-7.
- Granderath FA, Granderath UM, Pointner R. Laparoscopic revisional fundoplication with circular hiatal mesh prosthesis: the long –term results. World J Surg. 2008;32(6):999-1007.
- Frantzides CT, Madan AK, Carlson MA, Stavropoulos GP. A prospective, randomized trial of laparoscopic polytetrafluoroethylene (PTFE) patch repair vs simple cruroplasty of large hiatal hernia. Arch Surg. 2002;137(6):649-52.
- Eypasch E, Williams JL, Wood-Duaphinee S, Ure BM, Schmulling C, Neugebauer E, et al. Gastrointestinal quality of life index: development, validation and application of a new instrument. Br J Surg. 1995;82(2):216-22.
- Catarci M, Gentileschi P, Papi C, Carrara A, Marrese R, Gaspari AL, et al. Evidence-based appraisal of antireflux fundoplication. Ann Surg. 2004;239(3):325-37.
- Faria R, Bojke L, Epstein D, Corbacho B, Sculpher M. REFLUX trial group. Cost-effectiveness of laparoscopic fundoplication versus continued medical management for the treatment of gastro-oesophageal reflux disease based on long-term follow-up of the REFLUX trial. Br J Surg. 2013;100(9):1205-13.
- Broeders JA, Rijnhart-de Jong HG, Draaisma WA, Bredenoord AJ, Smout AJ, Gooszen HG. Ten-year outcome of laparoscopic and conventional nissen fundoplication: randomized clinical trial. Ann Surg. 2009;250(5):698-706.
- Zornig C, Strate U, Fibbe C, Emmermann A, Layer P. Nissen vs Toupet laparoscopic fundoplication. A prospective randomized study of 200 patients with and without preoperative esophageal motility disorders. Surg Endosc. 2002;16(5):758-66.
- Kellokumpu I, Voutilainen M, Haglund C, Färkkilä M, Roberts PJ, Kautiainen H. Quality of life following laparoscopic Nissen fundoplication: assessing short-term and long-term outcomes. World J Gastroenterol. 2013;19(24):3810-8.
- Hashemi M, Peters JH, DeMeester TR, Huprich JE, Quek M, Hagen JA, et al. Laparoscopic repair of large type III hiatal hernia: Objective follow-up reveals high recurrence rate. J Am Coll Surg. 2000;190(5):553-60.
- Stylopoulos N, Gazelle GS, Rattner DW. Paraesophageal Hernias: Operation or Observation? Ann Surg. 2002;236(4):492-500.
- Kohn G, Price R, Demeester SR, Zehetner J, Muensterer O, Awad Z, et al; the SAGES Guidelines Committee. Society of American Gastrointestinal and Endoscopic Surgeons.Guidelines for the Management of Hiatal Hernia. 2011.
- Nason KS, Luketich JD, Awais O, Abbas G, Pennathur A, Landreneau RJ, et al. Quality of life after collis gastroplasty for short esophagus in patients with paraesophageal hernia. Ann Thorac Surg. 2011;92(5):1854-60.
- Edye M, Salky B, Posner A, Fierer A. Sac excision is essential to adequate laparoscopic repair of paraesophageal hernia. Surg Endosc. 1998;12(10):1259-63.
- Zehetner J, Demeester SR, Ayazi S, Kilday P, Augustin F, Hagen JA, et al. Laparoscopic versus open repair of paraesophageal hernia: the second decade. J Am Coll Surg. 2011;212(5):813-20.
- Linke GR, Gehrig T, Hogg LV, Göhl A, Kenngott H, Schäfer F, et al. Laparoscopic mesh-augmented hiatoplasty without fundoplication as a method to treat large hiatal hernias. Surg Today. 2014;44(5):820-6.
- Dallemagne B, Kohnen L, Perretta S, Weerts J, Markiewicz S, Jehaes C. Laparoscopic Repair of Paraesophageal Hernia. Long-term Follow-up Reveals Good Clinical Outcome despite High Radiological Recurrence Rate. Ann Surg. 2011;253(2):291-6.
- Targarona EM, Grisales S, Uyanik O, Balague C, Pernas JC, Trias M. Long-term outcome and quality of life after laparoscopic treatment of large paraesophageal hernia. World J Surg. 2013;37(8):1878-82.
- Obeid NM, Velanovich V. The choice of primary repair or mesh repair for paraesophageal hernia. A decision analysis based on utility scores. Ann Surg. 2013;257(4):655-64.
- Antoniou SA, Koch OO, Antoniou GA, Pointner R, Granderath FA. Mesh reinforced hiatal hernia repair: A review on the effect on postoperative dysphagia and recurrence. Langenbecks Arch Surg. 2012;397(1):19-27.
- Chilintseva N, Brigand N, Meyer C, Rohr S. Laparoscopic prosthetic hiatal reinforcement for large hiatal hernia repair. J Visc Surg. 2012;149(3):e215-20.
- Antoniou SA, Antoniou GA, Koch OO, Pointner R, Granderath FA. Lower recurrence rates after mesh-reinforced versus simple hiatal hernia repair: a meta-analysis of randomized trials. Surg Laparosc Endosc Percutan Tech. 2012;22(6):498-502.
- Champion JK, Rock D. Laparoscopic mesh cruroplasty for large paraesophageal hernias. Surg Endosc. 2013;17(4):551-3.
- Priego P, Ruiz-Tovar J, Pérez de Oteyza J. Long-term results of giant hiatal hernia mesh repair and antireflux laparoscopic surgery for gastroesophageal reflux disease. J Laparoendosc Adv Surg Tech A. 2012;22(2):139-41.
- Carlson MA, Condon RE, Ludwig KA, Schulte WJ. Management of intrathoracic stomach with polypropylene mesh prosthesis reinforced transabdominal hiatus hernia repair. J Am Coll Surg. 1998;187(3):227-30.
- Amid PK. Shrinkage: fake or fact? In: Schumpelick V, Nyhus LM, editors. Meshes: benefits and risks Springer Berlin, 2004; pp 198–206.
- Feigal DW. Premarket approval of saline filled silicone prosthesis: United States-2000. Rockville, Maryland: Center for Device and Radiological Health, Food and Drug administration. 2000.
- The Silicones. In: Falcon J, editor. Encyclopedia of Polymer Science and Engineering. Wiley New York, 1989.
- Brandalise A, Aranha NC, Brandalise NA. The polypropylene mesh in the laparoscopic repair of large hiatal hernias: technical aspects. Arq Bras Cir Dig. 2012;25(4):224-8.